Murine VEGF-A ELISA kit
Product Specifications
- Catalogue N°
- 660.140.096 - 1 x 96 (pre-coated)
660.140.192 - 2 x 96 (pre-coated) - Assay Range
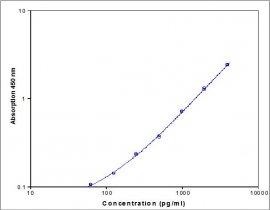
- 62.5 pg/ml - 4000 pg/ml
- Sensitivity
- 20pg/ml
- Target species
- Murine
- Specificity
- Recognizes both natural and recombinant murine VEGF-A
- Incubation
- 4h30
- Sample Type
- Cell culture supernatant and serum tested, other body fluids suitable
- Sample Size
- 50 µl
- Kit Content
- Diaclone ELISA Kits include pre-coated strip plates, biotinylated secondary antibody, standards, controls (where applicable), buffers, streptavidin-HRP, TMB, stop reagents and a detailed procedure.
- Synonym
- VPF
Related products
BACKGROUND
Normal tissue function depends on a regular supply of oxygen through the blood vessels. Understanding the formation of blood vessels has become the focus of a major research effort throughout the last decade. Vasculogenesis in the embryo is the process by which new blood vessels are generated de novo from primitive precursor cells. Angiogenesis is the process of new blood vessel formation from pre-existing vasculatures. It plays an essential role in development, normal tissue growth, wound healing, the female reproductive cycle (placental development, ovulation, corpus luteum) and also plays a major role in various diseases. Special interest is focused on tumor growth, since tumors cannot grow more than a few millimeters in size without developing a new blood supply. This process is described as tumor angiogenesis which is also essential for the spread and growth of tumor cell metastasis.
One of the key molecules for angiogenesis and for the survival of the endothelium is vascular endothelial growth factor (VEGF-A). It is a specific endothelial cell mitogen and a strong vascular permeability factor (VPF). VEGF-A is a heparin-binding glycoprotein, secreted as a homodimer of 45 kDa by many different cell types. VEGF-A also causes vasodilation through the nitiric oxide synthase pathway in endothelial cells and can activate migration in monocytes. Many different splice variants of VEGF-A have been described, but VEGF165 is the most predominant protein and anchors with its heparin binding domain to extracellular matrix and to heparin sulfate. During the past few years, several other members of the VEGF family have been cloned, including VEGF-B, -C- and -D. In terms of vascular angiogenesis, which mainly is regulated by VEGF-A, lymphangiogenesis is mainly regulated by VEGF-C and -D.
VEGF-A transcription is highly activated by hypoxia and by oncogenes like H-ras and several transmembrane tyrosine kinases, such as epidermal growth factor receptor and ErbB2. Together these pathways account for a marked upregulation of VEGF-A in tumors compared to normal tissues and are often of prognostic importance and relevance.
VEGF-A can be detected in both plasma and serum samples of patients, with much higher levels in serum. Extremely high levels can be detected in the cystic brain fluid of brain tumor patients or in ascites fluid of patients. Platelets release VEGF-A upon aggregation and may be another major source of VEGF-A delivery to tumors. Several other studies have shown that association of high serum levels of VEGF-A with poor prognosis in cancer patients may be correlated with an elevated platelet count. Tumors can release cytokines and growth factors that stimulate the production of megakaryocytes in the marrow and elevate the platelet count. This can result in another, indirect increase of VEGF-A delivery to tumors.
Furthermore, VEGF-A is implicated in several other pathological conditions associated with enhanced angiogenesis or enhanced vascular permeability. Examples where VEGF-A plays an important role are psoriasis and rheumatoid arthritis, as well as the ovarian hyperstimulation syndrome. Diabetic retinopathy is associated with high intraocular levels of VEGF-A, and inhibition of VEGF-A function may result in infertility by blockage of corpus luteum function. Direct demonstration of the importance of VEGF-A in tumor growth has been achieved using dominant negative VEGF receptors to block in vivo proliferation, as well as blocking antibodies to VEGF or to one of the VEGF receptors. Interference with VEGF-A function has therefore become of major interest for drug development to block angiogenesis and metastasis. More than 110 pharmaceutical companies world-wide are involved in the development of such antagonists. Their approaches include antagonists of VEGF-A or its receptors, selective tyrosine kinase inhibitors, targeting of drugs and toxins to VEGF receptors and gene therapy regulated by the same hypoxia pathway that controls VEGF-A production. Targeting the VEGF signalling may be of major therapeutic importance for many diseases and serves as a basis for the design of future (anti)-angiogenic treatments.
Version 3 - 07.19
For research use only
For any order, the purchaser acknowledges having read and accepted the terms and conditions described on the Diaclone website.



