Anti-Human TNF-α PE Conjugated
Product Specifications
- Catalogue N°
- 855.162.019 - 100 tests / 1ml
- Target species
- Human
- Specificity
- Recognises natural and recombinant human TNF-a
- Clone
- B-D9
- Application
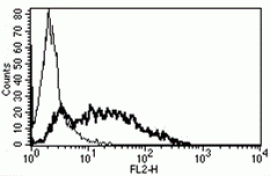
- Flow Cytometry
- Hybridoma
- Myeloma X63/AG.8653 x Balb/c spleen cells
- Immunisation
- Recombinant human TNF-a
- Quantity
- 100 Tests / 1ml (Discovery Size also available please enquire)
- Isotype
- Mouse IgG1 Kappa light chain
- Format
- Phosphate-buffered saline with 5% BSA and 0,1% sodium azide
- Storage
- Stable at +2-8°C for six months after reconstitution. DO NOT FREEZE.
- Working Dilution
- Reconstitute with 1ml of deionized water. It is recommended to establish the optimal dilution according to the system being used. Use 10 μl of antibody to label 5.105 cells
- Synonym
- TNF-a
- TNF-alpha
References
- Arduise, C. et al., J. Immunol.,2008; 181(10): 7002-7013. - Pubmed link
Related products
- 855.150.000 - Anti-Human TNF-α Azide Free
- 855.161.019 - Anti-Human TNF-α FITC Conjugated
- 879.090.001 - Anti-Human TNF-α Capture Antibody
- 879.090.002 - Anti-Human TNF-α Detection Antibody
- 860.040.048 - Murine TNF-α ELISA Kit
- 865.000.048 - Rat TNF-α ELISA Kit
- 950.090.048 - Human TNF-α ELISA Kit
- 851.570.001 - Human TNF-α ELISA Set
- 872.010.001 - Rat TNF-α ELISA Set
- 856.041.001PC - Human TNF-α ELISpot Kit
- 856.041.001 - Human TNF-α ELISpot Set
- 867.021.001 - Rat TNF-α ELISpot Set
- 867.021.001PC - Rat TNF-α ELISpot Kit
- 869.050.010 - Human TNF-α ELISpot Pair
- 871.030.010 - Rat TNF-α ELISpot Pair
- 855.330.000 - Anti-Human TNF-α Azide Free
 Sodium azide is a poisonous and hazardous substance, handle and dispose of properly
Sodium azide is a poisonous and hazardous substance, handle and dispose of properly
BACKGROUND
Tumor Necrosis Factor (TNFa), also known as cachectin, is a polypeptide cytokine produced by monocytes and macrophages. It functions as a multipotent modulator of immune response and further acts as a potent pyrogen. TNFa circulates throughout the body responding to stimuli (infectious agents or tissue injury), activating neutrophils, altering the properties of vascular endothelial cells, regulating metabolic activities of other tissues, as well as exhibiting tumoricidal activity by inducing localized blood clotting. TNFa also inhibits lipoprotein lipase activity resulting in cachexia, a physical wasting condition. Activation of B-cells by the Epstein Barr virus can be inhibited by TNFa. Due to its varied actions throughout the immune system, TNFa may play a role in the pathogenesis of many disease states.
TNFa production is mediated by the action of lymphokines and endotoxins on the macrophage. Purified monocytes produce TNFa within four hours of stimulation by recombinant IL-2 and there is some in vitro evidence to suggest that TNFa is expressed at high levels and with prolonged kinetics in T cells stimulated by both CD2 and CD28. Secretion of TNFa is enhanced by gamma interferon. TNFa then induces or enhances the specific production of Class I MHC antigen, GM-CSF, and IL-1. Recent evidence has suggested an intracellular role for this peptide.
TNFa may play a significant role in the pathogenesis of inflammatory disease of the joints and other tissues. Chin et al. found that TNFa, along with IFNg and IL-1 increased cell surface expression of ICAM-1 on synovial fibroblasts. Alvaro-Garcia et al. reported that TNFa stimulates synovial proliferation.
Waage et al. found that increased levels of TNFa in patients with septicemia and meningococcal disease correlated with fatal outcome. Scuderi et al. suggest that increased levels of this cytokine may play a role in the host defense mechanism against parasitic infections. Girardin et al. reported that increased serum TNFa levels correlated with the number of risk factors involved in children with gram-negative sepsis and purpura fulminians. Elevated levels of TNFa were also found in individuals suffering from myocarditis.
Role for TNFa in the pathogenesis of AIDS has also been pointed out. Alveolar macrophages (AM) from HIV positive individuals with opportunistic lung infections have been shown to spontaneously produce higher levels of TNFa in vitro than those HIV positive individuals without infection and HIV negative controls. Krishnan et al.report that higher TNFa production by AM was associated with lower counts of pneumocystis carinii in broncheoalveolar lavage fluid, indicating that TNFa may play a role in the control of this infection in AIDS. Israel-Biet et al.also reported in in vitro studies, that AM that express HIV (p24+) released significantly higher levels of TNFa than p24- alveolar macrophages and controls. Reddy et al. found persistently elevated levels of circulating TNFa in HIV seropositive individuals and suggest a possible involvement of this cytokine in the development of AIDS.
Measurement of TNFa levels has also been shown to be useful in transplant research, where Maury et al.and McLaughlin et al.. Both reported TNFa to be markedly elevated in renal allograft rejection episodes. Recent evidence has been presented on increased TNFa levels in Bone Marrow Transplant (BMT). BMT patients with major transplant related complications such as interstitial pneumonitis and severe acute graft-versus - host disease had TNFa levels significantly increase over controls.
Version 10 - 06.19
For research use only
For any order, the purchaser acknowledges having read and accepted the terms and conditions described on the Diaclone website.



